Thanks for tuning in to part 2 of this two-part feature of VTE. The first segment, which focuses on DVTs, can be found here!
So now we are going to focus on the more critical version of a VTE. Yes, DVTs are critical. They are important to assess and treat quickly. But they are less acutely life threatening than a pulmonary embolism. Let’s take a closer look at PEs and how they are relevant to rehab clinicians.
A pulmonary embolism is something (usually a blood clot) that moves through the vasculature and eventually impedes a vessel that supplies a portion of the lung. This sudden blockage is typically a clot, but can potentially be several different things such as fat, bone, soft tissue, air, etc. You may be thinking, “How would any of those things get in to an artery and cause a blockage in the lungs?” Well, through external means. People who undergo surgeries are at an increased risk for PE because the act of cutting through tissue can introduce those elements in to the arterial system. Crush injuries can also introduce tissues in to the vasculature that doesn’t belong there. If these insults are relatively small enough to pass through our peripheral vasculature, they can make it back to the heart, and are then pumped into the pulmonary arterial supply. The emboli are actually pretty large because they are typically formed in the deep leg veins (they start as DVTs) which then dislodge and travel back to the heart through our relatively larger veins. However, vessels in the lungs get very small very quickly (they have to be capillary sized to perform oxygen and CO2 diffusion at the site of alveoli), so blockages are likely to happen here.

Just like with DVTs, PEs have a particular set of symptoms that can be quantified on a couple different scales. Like we talked about in the first post, the Wells’ Criteria has a scale for PE risk assessment. It works pretty much the same as the DVT risk assessment and the criteria are similar. As you can see here, I’ve set the tool for an assessment of a patient with COVID-19 with a typical presentation. However, this score could also be given to a patient after a joint replacement as the assessment would look very similar. The bonus to using the Wells’ Criteria is that it factors in the clinical gestalt. We mentioned this briefly in the last post. Your gestalt has been researched thoroughly and it has actually been shown that clinical gestalt (the feeling that something is amiss) is actually the single most important clinical tool in determining the need for further workup. If all your tools are telling you everything is fine, but you feel something is wrong, something is probably wrong, and there is research to back that up.

Another tool becoming more commonly used is the Revised Geneva Tool (rGeneva). Meta-Analysis outcomes have shown that the rGeneva is actually just as accurate in predicting risk of PE being present as the Wells’ criteria. The difference between the tools is that the rGeneva stratifies risk across three categories (low, medium, and high). The original version of the rGeneva utilized outcomes of ABGs and other assessments that take precious time to order, measure, and interpret, so the tool was revised to be done with only a physical patient assessment. This tool does not weigh clinician gestalt, so it is felt to have less clinical strength than the Wells’ Criteria, however, still very valid and useful.
How to inform your gestalt
There are some clinical factors that should be informing your gestalt when it comes to pulmonary emboli, some physical assessment pieces that should take you down the road of either ruling this out or calling 911. In a patient who has been immobilized or with recent surgery, the most obvious signs would include:
- Shortness of breath at rest or with minimal exertion
- (believe it or not, shortness of breath is NOT ALWAYS PRESENT in patients with pulmonary emboli!)
- Tachycardia and tachypnea
- Significant decline in functional status
- Confusion
- Chest pain with breathing (pleuritic pain) (65% of patients)
- Hemoptysis (65% of patients)
Other less common clinical factors may include:
- Diminished lung sounds and crackles unilaterally (up to 37% of patients)
- Pleural friction rub
- Pleural effusion
- Audible breath sounds (without auscultation)
- Sense of Impending Doom
- Coughing or wheezing
- Arm or leg swelling
If these factors are coming up in your physical exam, patient interview, or functional assessment, you should be very concerned and doing some deeper digging, as well as probably contacting a physician right away to report findings.
So, since I seem to have a patient or two with just about everything, yes, I’ve had several patients where I have identified a new PE. Two were post-surgical (cervical spine fusion and knee replacement), one was severely ill with end-stage COPD, and one was young but immobilized long term due to primary progressive MS. Two of these four died upon reaching the hospital. The patient with the cervical spine fusion had three PEs on his imaging but made it! The one with COPD didn’t return home. All of them were confused, some severely confused, three had a sense of impending doom, all four had abnormal lung auscultation, three had leg swelling. No one had a cough or difficulty breathing.
What happens next?
So we think our patient may have a PE. We’ve called the doctor or sent them to emergency. What happens to them now? They will get some blood work done (a D-dimer and ABGs) and some imaging studies performed. Likely, a CT angiography will be performed. I don’t know if you guys have ever seen an angiogram for a PE, but the images produced are just completely amazing and the clots tend to be pretty obvious. In the image below, image a is a slice of the pulmonary vasculature. The vessel heading off to the right (superior vena cava) is occluded with a large embolism (the shaded tubular shaped area within the white space). And image b is just incredible, but all those tiny dark spots within the white space (the vessels) are subsequent emboli.
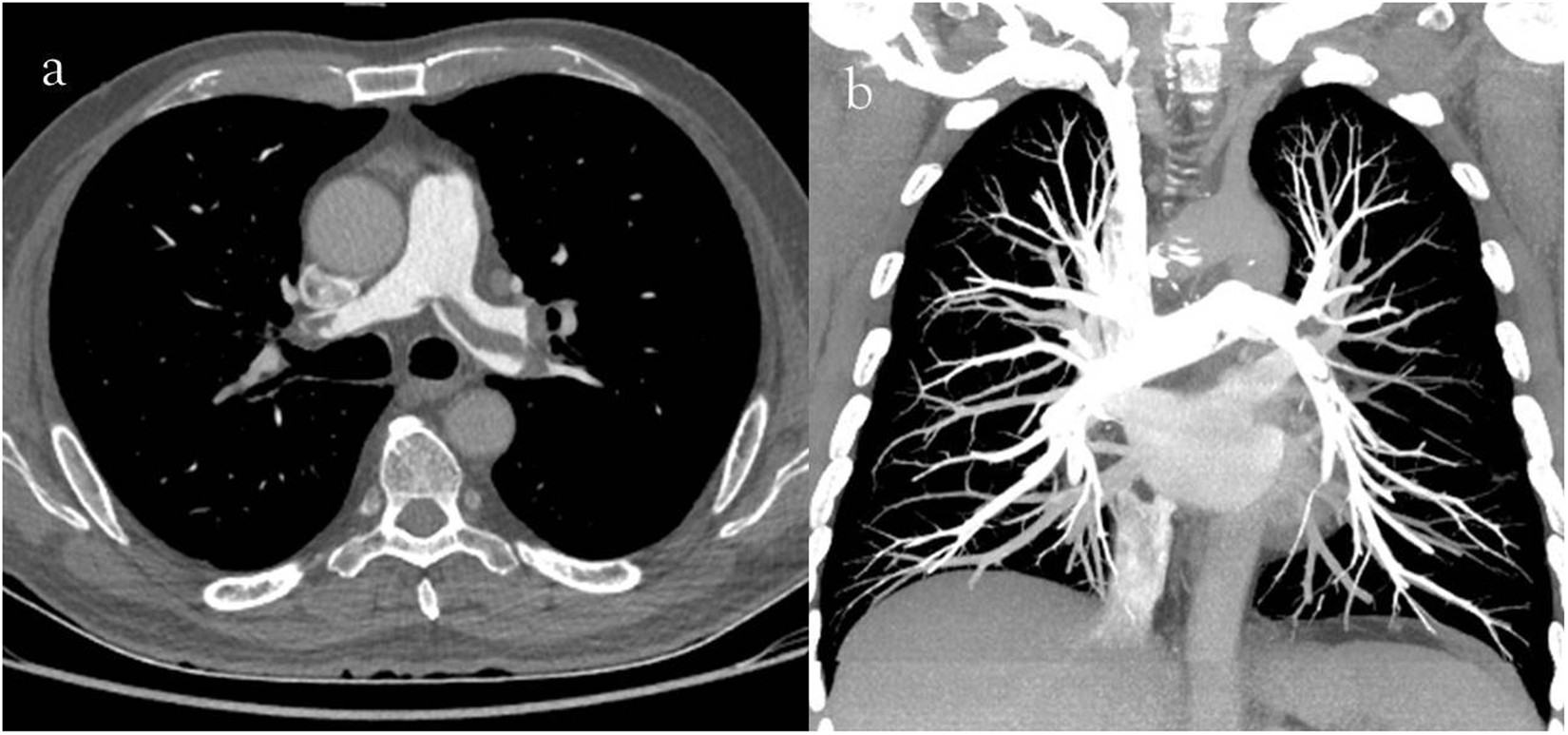
This is obviously a large embolus. Emboli that are very large like this one present in the large vasculature can cause major damage as they block such a large vessel which means every other lower-order vessel is now also blocked. These type of emboli tend to emanate from the deep leg veins and are more common in patients who have heart rhythm issues (another reason beta-blockers are so important!). You can see in the assessment tools that there are other factors that increase the risk for developing PEs:
- Cancer
- Cardiovascular disease
- Disorders that affect clotting
- Pregnancy
That last one is interesting. While living in West Michigan, I regularly encountered patients who had a condition called Factor V. People of European Caucasian decent have an increased risk of carrying a genetic mutation that results in Factor V Leiden, a clotting factor malfunction. This results in an increase in the risk of clotting including DVTs and PEs. There are thousands of clotting disorders out there that increase the risk of clotting.
Preventing PEs
Sometimes there just isn’t much you can do. PEs are one of those things that can pop up over night and have no symptoms until someone is so far gone that you aren’t sure if they are coming back. I’ve had a patient with no symptoms one day, and the next day I had to send him straight to emergency. There are some things we can do to lower the risk, and this list is going to look pretty similar to the list for DVT prevention:
- Promote mobility programs
- Promote anticoagulation adherence
- Promote rhythm drug adherence
- Graduated compression stockings
- Frequent position chages
- Educate patient on signs and symptoms
- Alternating pressure/compression devices
- Smoking cessation
- Weight loss

Does COVID-19 increase the risk for PEs?
As part of DIC (disseminated intravascular coagulopathy), the quick answer is yes. For more on DIC, see the first post in the VTE series. A new study found that 23% of patients in the ICU with COVID-19 had PEs. And I mean several PEs, not just one or two. The presence of PEs was difficult to determine due to the patient’s COVID-19 symptoms so angiography was typically performed much later than usual. The presence of PEs was determined to increase the need for and length of mechanical ventilation.
I wanted to show you some imaging for comparison. You saw the image above of the PEs in otherwise normal lungs. Below you will see what lungs look like when they not only have several PEs, but all the sequelae of COVID-19 including bronchiectasis, ground glass opacities, consolidation, and actual architectural breakdown. This particular set of lungs was thought to have a fluid volume of between 25-50%. You don’t even have to know the specifics of what all of that means to look and this set of images and realize this person is in rough shape.
Other studies have shown that between 20-30% of all patients with COVID-19 may have PEs whether they are in the ICU or not. In Detroit, doctors diagnosed 51% of the COVID-19-related PEs in the emergency department and 72% of them did not require ICU admission. However, what this can be interpreted as meaning is that even if your patient isn’t critically ill with COVID-19, their risk of having a PE is still very high.
So if you are seeing patients in the outpatient, home, or sub-acute settings, not only does their risk of having a PE or DVT increase simply because they had COVID-19, but the risk stays high and hospitalization for COVID-19 is not a factor is their PE risk. Even in patients who were on prophylactic treatment for clotting, almost a quarter of them still developed PEs. Rehab clinicians need to be watching out for the signs and symptoms of PE and DVT for any and all patients who are being treated after COVID-19 diagnosis or suspected COVID-19.
Have you ever spotted a PE? Tell me about it in the comments!
Follow my blog for more!
References:
Ceriani, E., Combescure, C., Le Gal, G., Nendaz, M., Perneger, T., Bounameaux, H., Perrier, A., & Righini, M. (2010). Clinical prediction rules for pulmonary embolism: a systematic review and meta-analysis. Journal of thrombosis and haemostasis : JTH, 8(5), 957–970. https://doi.org/10.1111/j.1538-7836.2010.03801.x
Grillet, F., Behr, J., Calame, P., Aubry, S., Delabrousse, E. (2020). Acute pulmonary embolism associated with COVID-19 pneumonia detected by pulmonary CT angiography. Radiography. ePub online ahead of print. Retrieved from https://doi.org/10.1148/radiol.2020201544
Le Gal, G. (2020). Geneva score (revised) for pulmonary embolism. Retrieved from mdcalc.com/geneva-score-revised-pulmonary-embolism#creator-insights
Penaloza, A., Verschuren, F., Meyer, G., Quentin-Georget, S., Soulie, C., Thys, F., & Roy, P. M. (2013). Comparison of the unstructured clinician gestalt, the wells score, and the revised Geneva score to estimate pretest probability for suspected pulmonary embolism. Annals of emergency medicine, 62(2), 117–124.e2. https://doi.org/10.1016/j.annemergmed.2012.11.002
Poyiadji, N., Cormier, P., Patel, P.Y., Hadied, M.O., Bhargava, P., Khanna, K., Nadig, J., Keimig, T., Spizarny, D., Reeser, N., Klochko, C., Peterson, E.L., Song, T. (2020). Acute pulmonary embolism and COVID-19 (a research letter). Radiology. Retrieved from https://pubs.rsna.org/doi/pdf/10.1148/radiol.2020201955
Stein, P. D., Beemath, A., Matta, F., Weg, J. G., Yusen, R. D., Hales, C. A., Hull, R. D., Leeper, K. V., Jr, Sostman, H. D., Tapson, V. F., Buckley, J. D., Gottschalk, A., Goodman, L. R., Wakefied, T. W., & Woodard, P. K. (2007). Clinical characteristics of patients with acute pulmonary embolism: data from PIOPED II. The American journal of medicine, 120(10), 871–879. https://doi.org/10.1016/j.amjmed.2007.03.024
Wells, P. (2020). Wells’ criteria for pulmonary embolism. Retrieved from https://www.mdcalc.com/wells-criteria-pulmonary-embolism
Follow @DoctorBthePT on Twitter for regular updates!





11 thoughts on “Venous Thromboembolism (VTE) Part 2: PEs”