This is the first post in a two-part series about understanding and interpreting arterial blood gases! If you want the rest of the post, you’ll have to check back next week!
I’m getting real science-y again!
So, just a heads up: my undergraduate degree was in chemistry. Kind of by accident, I took a lot of chemistry courses. I know that sounds a bit weird, but I had an odd schedule and chemistry classes filled the gaps. I didn’t even necessarily like them, but I did gain a lot of understanding when it came down to organic chemistry and physiological concepts. When it came time to learn about acid-base buffers, I was ahead of the game. When it came time to apply that to the human body, I was all over it. That’s how I view arterial blood gases. It’s like a chemistry homework question from undergrad. You just have to apply the same conceptual framework every time and you will figure it out. So, I’m going to teach you that conceptual frame work, step by step. I promise, it won’t be that hard!
Arterial blood gases (ABGs) are performed when it is suspected that a patient may be in alkalosis or acidosis (depending on the circumstances). It is a relatively painful arterial puncture (…so I’m told. I’ve never had one, but all my patients say it hurts much more than venipuncture). The arterial blood is oxygenated (unlike venous blood), so it shows you a bunch of information about how the acid-base buffer system in the human body (the BiCarb Buffer System) is functioning. These tests are regularly performed on people who have chronic lung, heart, or kidney disease, because all three of these organ systems are intricately intertwined and have significant impact on the oxygenation of tissues. However, there are some other diagnoses that will result in a patient requiring ABGs to be checked and we will talk about some of those here, too!
H+ + HCO3- < — > H2CO3 < — > CO2 + H2O
The above equation basically runs our body chemistry. We want to hang out around the middle. Humans have a VERY small window in which we can properly function, but we’ll talk more about that later. For the basic purposes of this post, we will say that humans exhale CO2 and inhale oxygen. We know there are lots of other gases involved, but we are going to focus on those for now. So you can imagine, if we exhale too much, we lose too much CO2 and we fall too far toward the left. If we don’t exhale enough, we accumulate too much CO2 and we fall too far toward the right. CO2 has a higher affinity for hemoglobin than oxygen (that good old oxygen-hemoglobin dissociation curve from physiology soooo many years ago…) so we have to focus on CO2. I know, all of this sounds a bit overwhelming. There is a lot to know, but I promise, I will break it down and make it easy for you!
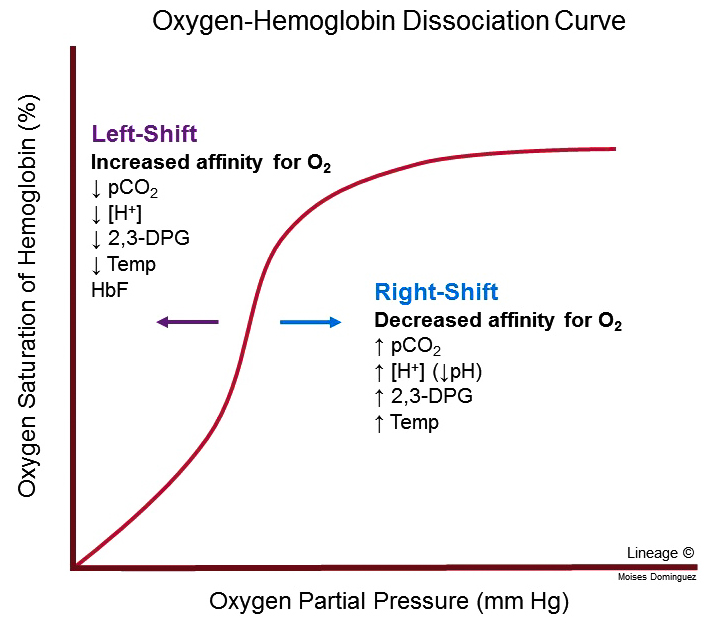
Basically, what this O2-HbG dissociation curves shows us is that the more oxygen we have, the easier it is to bind more oxygen. But as we lose oxygen and bind more CO2, it becomes easier to lose oxygen and bind more CO2. This is called the Bohr effect and, as you can see, it can hit a critical point. You can see that it is also effected by temperature and pressure. You can probably infer that profound anemia would also impact this curve. You can read more here about how the amount of hemoglobin affects your oxygen levels.
Chemistry is a really gray area in that it does its best to estimate amounts, but the reality is that, in a living system, they are estimates in time. Think about water. At any given time, you have liquid water, H2O, as well a gaseous hydrogen and oxygen molecules floating around binding and unbinding with each other in all different combinations (like H2O2 – hydrogen peroxide which is very volatile) in the same container. Water is a very stable molecule, but many bodily substances are not. Let’s talk about acidosis and alkalosis. We need to define a couple terms in case you aren’t as fresh on your chemistry:
pH. Typically thought of as “how acidic or basic something is”. A low pH indicates acidic, a high pH indicates basic. Normal human pH is 7.34 to 7.45.
Acidosis. When too many hydrogen ions [H+] build up in the body in some form (carbonic acid or lactic acid). Hydrogen ions are the basic unit of an acid. Body pH would be below 7.35.
Alkalosis. When too much bicarbonate [HCO3-] builds up in the body. Bicarbonate is a base. Body pH would be above 7.45.
Now that you’ve got the basics, let’s do some application.
Respiratory Acidosis. Results from hypoventilation leading to increased CO2 in blood and decreased pH. Things that cause this condition include pretty much anything that decreases your ability to remove CO2 (diffusion and ventilation). That would include respiratory suppression from drug usage, Giullian-Barre Syndrome, a brain injury to respiratory center, or airway obstruction (acute or chronic).
However, this condition can also be caused by an increased oxygen metabolism without compensating with a change in ventilation. What might cause that? A large amount of tissue damage requiring a large amount of healing such as in burns or sepsis (like COVID-19). When it presents acutely, respiratory acidosis is a medical emergency and requires immediate medical intervention. Acute-on-chronic respiratory acidosis would also require emergency medical intervention.

Wait… Did Doctor B just say that a medically emergent condition can be chronic? Yep, I did. Respiratory acidosis can be chronic in patients with COPD and some other obstructive airway conditions. Their bodies get so used to being oxygen deprived that they no longer compensate with an increased respiratory rate. Respiratory acidosis just becomes their normal state. Arterial blood gases then really come in to play to establish when someone becomes a CO2 retainer. This is a HUGE safety factor and one of the most important reasons rehab professionals need to be able to interpret ABGs. Please read this post on CO2 retainers and this post on oxygen saturation for more information.
Respiratory Alkalosis. This happens when hyperventilation results in the removal of too much CO2 which results in increased pH. Respiratory alkalosis is rarely life threatening (unless the patient is also in acute respiratory distress) and can be easily treated. Panic attacks are sometimes characterized with these symptoms, and can result in people passing out, which effectively stabilizes their respiratory rate and they return to normal.
This is one of the complications for patients who have COVID-19. They are in respiratory distress due to low oxygen levels, but then can also push too far in to alkalosis by breathing too quickly trying to get more oxygen (which won’t be effective due to parenchymal damage). This can also be a complication in the process of recovering from COVID-19, as patients can demonstrate oxygen starvation resulting in similar behavior. Treatment is fairly simple if the patient isn’t in ARDS: reduce the respiratory rate or use a rebreather. These treatments can be achieved in a few different ways:
- Reducing respiratory rate could be achieved by breathing control, diaphragmatic breathing, or increasing oxygen titration (as able). Sometimes distraction, prayer, meditation, biofeedback, or education can help with this also.
- Using a rebreather is probably what you think of when you see someone breathing in to a paper bag. We, of course, would use medical equipment for this type of thing, with different rebreather masks available, but the concept is the same. The patient breathes in some of the CO2 they exhaled in to the bag/mask and the concentration of CO2 in their blood increases.
When treating respiratory alkalosis, you have to monitor to ensure your patient doesn’t swing the opposite direction in to respiratory acidosis by increasing their CO2 levels too much.
Metabolic acidosis. This results when the body produces too much acid and the kidneys cannot remove it quickly enough which results in a decreased pH. As with respiratory acidosis, this is a medical emergency, and can result in coma or death. Metabolic acidosis can be caused by several factors: rhabdomyolysis, starvation, exercise bulimia, aspirin (abbreviated ASA = acetyl-salicylic acid) overdose, etc… as these processes result in acid build up. Oddly, overuse of the laxative polypropylene glycol (MiraLax) that ALL of your patients are on can also cause metabolic acidosis due to the chemical breakdown of this substance. I know that sounds weird, because you’d think if they were getting rid of all that stomach acid, they would become alkaline, but that isn’t the case with this specific agent. MiraLax is metabolized in to an acid. Other causes may include kidney failure and acute/chronic ethanol intoxication, but many causes are possible and it is difficult to detect without ABGs.

However, just like with respiratory acidosis, metabolic acidosis can also become chronic. This can happen in patients who have uncontrolled Type 2 Diabetes Mellitus. We just have a different name for it (ketoacidosis). Ketoacidosis can be acute as well, so you still need to be calling for medical help. Thankfully, there is something you can do for these patients:
- If you happen upon someone who is in metabolic acidosis, you can help them temporarily (long enough to call 911 and get an ambulance to them) as this condition can be compensated using hyperventilation! This works by expelling the CO2 making up carbonic acid in their bodies to slightly offset the acid levels. Like I said, this is temporary and only buys time. It is NOT a cure! Metabolic acidosis will persist until treated.
Metabolic alkalosis. The removal of too much acid (or too many H+ ions) from the body which increases pH. Metabolic alkalosis can be caused by vomiting, diarrhea, severe diuresis, dehydration, or, in rare cases, loss of sodium in sweat glands solely associated with cystic fibrosis. Metabolic alkalosis cannot last long if the kidneys are well functioning, as the kidneys will compensate by retaining more H+ ions and Na+ ions. This condition typically presents with low blood pressure, low blood volume, elevated heart rate, and lethargy. Patients may be significantly orthostatic. However, it can also present with hypervolemia in the case of heart failure patients in fluid overload depending on the electrolyte factors involved and the kidney function impairments.
This condition can also be compensated in the same way respiratory alkalosis can be compensated: with use of a rebreather or by reducing the respiratory rate.
As it so happens, though, I see people with metabolic alkalosis ALL THE TIME who need medical intervention. My typical caseload involves people with mulitple complex chronic diseases. For many them, their kidneys are NOT functioning properly. For most, medical intervention takes the form of IV fluids with different levels of sodium (Na+), potassium (K+), or lactic acid (Ringer’s Lactate solution). For those with heart failure, they can actually be in fluid overload but not have any usable fluid so be exhibiting the symptoms of hypovolemia at the same time. This becomes a delicate dance of giving them enough usable fluid while simultaneously pulling off the unusable fluid, which has to be done under close observation in the inpatient setting.
OK, so now that you’ve got the basics, I’m going to let you marinate on that for a while. In Part II of this post, we will talk about compensated conditions, how to read ABGs, and how to tell what condition your patient is in. This will help you understand when it is safe to treat them based on these values. Keep checking back!
How often do you get the chance to read ABGs before the ordering provider? How do you use that information? Tell me about it in the comments!
Kaufman, D. (2020). Clinical Education: Interpretation of Arterial Blood Gases (ABGs). American Thoracic Society. Retrieved from http://www.thoracic.org/professionals/clinical-resources/critical-care/clinical-education/abgs.php
Lewis, J. (2020). Acid-Base Disorders. Merck Manual Professional Version. Retrieved from https://www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/acid-base-regulation-and-disorders/acid-base-disorders
Follow @DoctorBthePT on Twitter for regular updates!





7 thoughts on “ABGs (Part 1)”